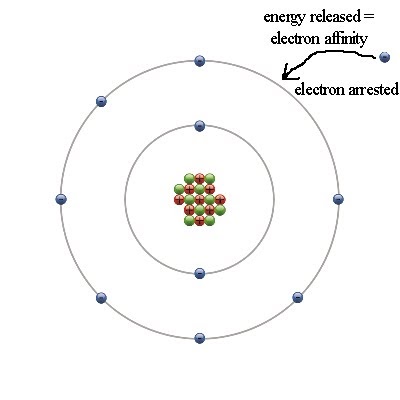

For example, if the atomic radius is high, a low amount of energy is required since the outermost electron is located far from the nucleus. However, the value also depends on some other facts. If there is an unpaired electron, it requires a lower energy. If all electrons are paired in an atom, it requires a higher energy. However, the energy required for this purpose depends on the type of element. This concept is associated with a neutrally charged atom since neutrally charged atoms are composed only of the original number of electrons that the element should be composed of. This reaction is essentially an endothermic reaction. Hence, the first ionization energy is the energy required to discharge the highest energy electron from an atom. Therefore, this electron has the highest energy among other electrons of that atom. This outermost electron is located in the outermost orbital of an atom. First Ionization Energyįirst ionization energy is the amount of energy required by a gaseous, neutral atom to remove its outermost electron. For example, the first and second ionization energies describe the amount of energy required by an atom to remove one electron and another electron, respectively. Each and every chemical element has a specific ionization energy value since atoms of one element are different from atoms of another element. This is called the ionization energy because the atom gets a positive charge after the removal of an electron and becomes a positively charged ion. Ionization energy is the amount of energy needed by a gaseous atom in order to remove an electron from its outermost orbital. When the atomic number increases, the number of electrons they have in their outermost orbitals increases. The elements of the periodic table are arranged according to the ascending order of their atomic number. This is because the incoming electron is added to the outermost orbital of an atom. Second Electron Affinity: X (g) – + e – → X (g) -2Įlectron affinity shows periodic variation in the periodic table. Therefore, the first electron affinities are negative values and the second electron affinity values of the same species are positive values.įirst Electron Affinity: X (g) + e – → X (g) – This phenomenon is called an endothermic reaction. This is because the incoming electron is repelled by the other electrons. But if another electron is going to be added to this negative ion, energy should be given in order to proceed with that reaction. The addition of an electron to a neutral atom or a molecule releases energy. This can be represented by symbols as follows. This electron addition causes the formation of a negatively charged chemical species. Key Terms: Atom, Electron, Electron Affinity, Electron Gain Enthalpy, First Ionization Energy, Ionization Energy, Second Ionization EnergyĮlectron affinity is the amount of energy released when a neutral atom or a molecule (in the gaseous phase) gains an electron from outside.

What is the Difference Between Electron Affinity and Ionization Energy What are the Similarities Between Electron Affinity and Ionization EnergyĤ. – Definition, First Ionization, Second Ionizationģ. – Definition, Endothermic and Exothermic Reactions The main difference between electron affinity and ionization energy is that electron affinity gives the amount of energy released when an atom gains an electron whereas ionization energy is the amount of energy required to remove an electron from an atom. Ionization energy, on the other hand, is the amount of energy required to remove an electron from an atom. Electron affinity can be also known as electron gain enthalpy when the meaning is considered, but they are different terms since electron gain enthalpy describes the amount of energy absorbed by the surrounding when an atom gains an electron. Electron affinity is the amount of energy released when a neutral atom or molecule gain an electron.

Electron affinity and ionization energy are two such concepts in chemistry. There are many chemical concepts to explain the behavior of electrons. Main Difference – Electron Affinity vs Ionization EnergyĮlectrons are subatomic particles of atoms.
